Our clinical themes and academic work streams will form an integrated framework to develop technologies through our 5- step innovation cycle:
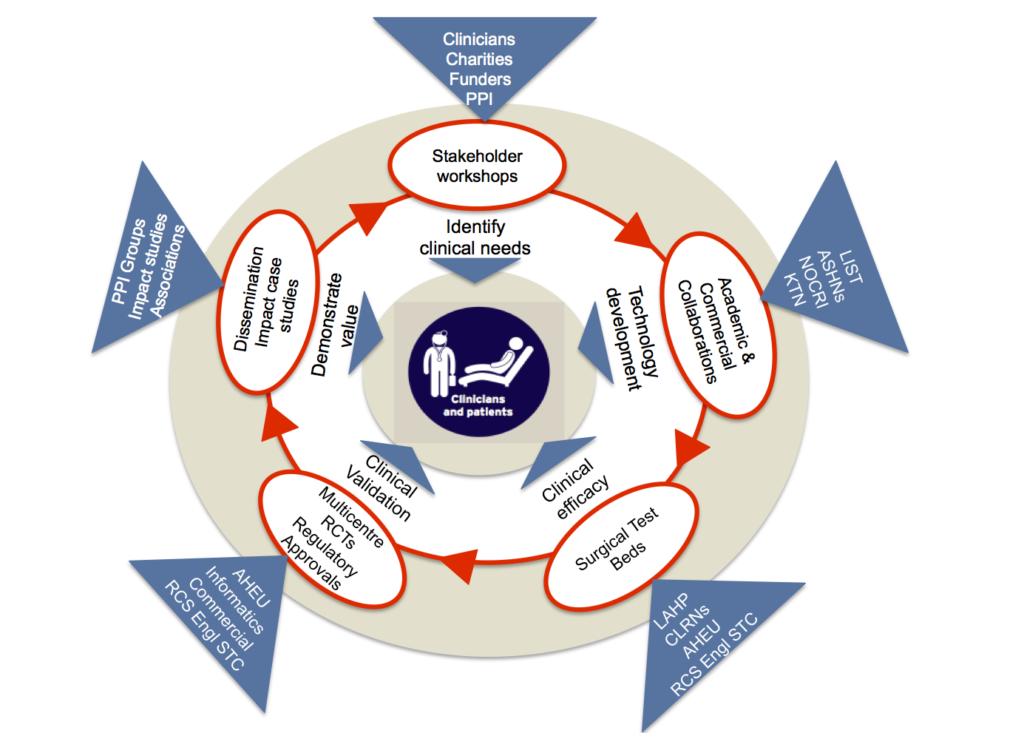
i) Identification of unmet clinical needs
This will involve hosting workshops to identify unmet clinical needs and “sandpit” events to identify projects to progress for funding. Patients and members of the public will be invited to multidisciplinary workshops, sandpit events and related advisory groups to ensure that the patient needs are included.
ii) Technology development
We will use pump-prime funding and “incubator packages”, which have proven effective in building research consortia; and our patient groups will be involved in the commissioning and prioritisation of new research.. This will also involve partnering with other networks to develop collaborative projects with support for early economic modelling, industry secondments and academic engagement.
iii) Clinical efficacy
Our Surgical Technology Testbed will be expanded to include neurosurgery, vascular and HPB surgery, to enable research into new models of surgical trials that will shorten the evaluation time and cost for industry. Our Health Economics work stream will provide detailed cost-effectiveness and resource utilisation models to support the business case for adoption. Similar technology evaluations will be initiated through our spoke sites.
iv) Validation
This will involve close working with the Leeds Royal College of Surgeons (RCS) Surgical Trials Centre and the Academic Unit for Health Economics to deliver clinical trials and cost effectiveness evaluation for industry. For innovation to have a positive impact on patient outcomes, we need to reduce the time it takes from idea to adoption; our patients groups will play a key part in this through their involvement in surgical technology evaluations.
v) Demonstration of value
We will provide expertise in Business Development and Commercialisation to ensure that projects progress towards clinic adoption. Dissemination is an important activity throughout, and we will use our links with patients and the public, professional associations, NIHR infrastructure, MedTech, procurement officers and commissioners to disseminate outputs.
Download Surgical MedTech Co-operative Brochure
